Website:
Depuy Synthes
Website:
Depuy Synthes
Group: Johnson & Johnson
Catalog excerpts

For the Attention of the Operating Surgeon: ZERO-P NATURAL™ PLATE INDICATIONS The ZERO-P NATURAL Plate is intended for anterior plate and screw fixation of the cervical spine. The plate and four screw system has been designed for use with structural allograft to provide stabilization as an adjunct to cervical fusion. Indications for use of this plate system include degenerative disc disease (defined as neck pain of discogenic origin with degeneration of the disc confirmed by history and radiographic studies), spinal stenosis, pseudarthrosis or failed previous fusion. The plate is intended for one level in the cervical spine, from C2 to T1. DESCRIPTION The ZERO-P NATURAL Plate and four screw system consists of an assortment of plates and screws to suit varying patient anatomy. The plates are available in sizes ranging from 6–12 to accommodate one level of fixation. The 3.0mm selftapping screws are available in lengths of 12mm, 14mm and 16mm. The screws are supplied separately and are available sterile packaged and non-sterile packaged as specified in Table 1. Table 1 Screws Catalog Number 04.617.812 04.617.814 04.617.816 04.617.812.02S 04.617.814.02S 04.617.816.02S Description 3.0mm Ti Cervical Spine Locking Screw 12mm 3.0mm Ti Cervical Spine Locking Screw 14mm 3.0mm Ti Cervical Spine Locking Screw 16mm 3.0mm Ti Cervical Spine Locking Screw 12mm - Sterile 3.0mm Ti Cervical Spine Locking Screw 14mm - Sterile 3.0mm Ti Cervical Spine Locking Screw 16mm - Sterile The ZERO-P NATURAL Plate has been designed for use with the corticocancellous (CC) Natural allograft spacer processed by the Musculoskeletal Transplant Foundation (MTF). Specific pairings of ZERO-P NATURAL Plates and CC Natural allograft spacers provided in Table 2 ensure a proper fit and height relationship of the allograft spacer to the plate. Table 2 Pairings of ZERO-P NATURAL Plates and MTF CC Natural Allograft Spacers Trial Spacer MTF CC Natural Allograft Spacer Includes standard and large footprints and parallel and lordotic sagittal profiles Height 6mm 7mm 8mm 9mm 10mm 11mm 12mm Use of an allograft spacer with height less than that of the plate may cause the plate to stress-shield the allograft spacer and may result in increased risks to the patient, such as, subsidence, poor spinal mechanics, loosening, malunion or nonunion. The plate retention arms are not intended to maintain disc height and are not designed to contact the vertebral body endplates. Use of an allograft spacer with a height that is too large for the selected plate may result in increased risks to the patient, such as, poor spinal mechanics, malunion, nonunion, or loosening. The MTF instructions for use should be followed for preparation of the CC Natural allograft spacer. Each ZERO-P This document is valid only on the date printed. If unsure of the print date, please re-print to ensure use of the latest revision of the IFU (available at www.e-ifu.com). The onus resides with the user to ensure that the most up-to-date IFU is used.
Open the catalog to page 1
NATURAL Plate is sterile packaged with an assembly tool, as shown in figure 1, to position the plate retention arms to accept the CC Natural allograft spacer. Once the plate and CC Natural allograft spacer are attached the assembly tool is removed. The plate and CC Natural allograft spacer are then simultaneously inserted using the aiming device or implant holder. Four screws are inserted through the plate into the adjacent vertebral bodies and locked to the plate. The surgical technique guide provides detailed instructions for implantation of the ZERO-P NATURAL Plate. 1. Zero-P Natural...
Open the catalog to page 2
• Mixing metals can cause corrosion. There are many forms of corrosion and damage and several of these occur on metals surgically implanted in humans. General or uniform corrosion is present on all implanted metals and alloys. The rate of corrosive attack on metal implant devices is usually very low due to the presence of passive surface films. Dissimilar metals in contact, such as titanium and stainless steel, accelerates the corrosion process of stainless steel and more rapid attack occurs. The presence of corrosion often accelerates fatigue and fracture of implants. The amount of metal...
Open the catalog to page 3
As necessary, remove adjacent-level implants that prevent ZERO-P NATURAL Plate from being implanted according to the surgical technique. • Caution: Placement of ZERO-P NATURAL Plate adjacent to a previous, multi-level fusion could result in increased loading. Supplemental fixation should be considered in cases where ZERO-P NATURAL Plate is placed adjacent to a previous, multi-level fusion. • Do not place a ZERO-P NATURAL Plate adjacent to existing implants if the adjacent level cannot be confirmed to be fused or where fusion has not occurred. • Important: Use imaging to verify final implant...
Open the catalog to page 4
PATIENT INFORMATION POSTOPERATIVE MOBILIZATION Post-operative care and the patient’s ability and willingness to follow instructions are among the most important aspects of successful bone healing. The patient must be made aware of the limitations of the implant, and instructed to limit and restrict physical activities, especially lifting and twisting motions and participating in any type of sports. The patient should understand that a metallic implant is not as strong as normally healthy bone and could loosen, bend and/or break if excessive demands are placed on it, especially in the...
Open the catalog to page 5All Depuy Synthes catalogs and technical brochures
-
2.0 mm LCP® Distal Ulna Plate
20 Pages
-
SYNFLATE SYSTEM
3 Pages
-
2.4 mm VA LCP™
4 Pages
-
Building on Success
16 Pages
-
HEALIX ADVANCE
4 Pages
-
RADIUS OF CURVATURE
3 Pages
-
Introducing The Variable Angle
12 Pages
-
HEALIX Anchor™ 3.4 mm
2 Pages
-
Small Battery Drive II
4 Pages
-
HEALIX ADVANCE
4 Pages
-
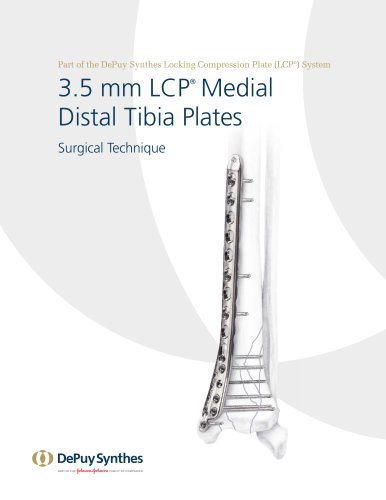
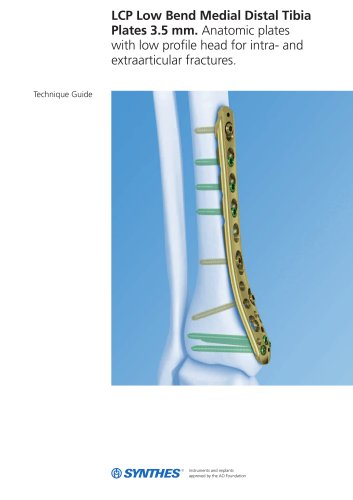
3.5 mm LCP™ Medial
15 Pages
-
Titanium Sternal Fixation System
34 Pages
-
MatrixRIB®FixationSystem
86 Pages
-
Mandible Trauma Solutions
2 Pages
-
Power line II
4 Pages
-
Concorde
28 Pages
-
LCP Intercarpal
31 Pages
-
LCS® COMPLETE™
2 Pages
-
Synthes TPLO.
8 Pages
-
SynFix-LR System
56 Pages
-
ATB Anterior Tension Band Plate
32 Pages
-
CONDUIT™
15 Pages
-
Brochure_FINAL
2 Pages
-
DePuy Synthes
81 Pages
-
Anspach
3 Pages
-
Orthopedic Foot Instruments
32 Pages
-
PINNACLE® Hip Solutions
12 Pages
-
Corail
24 Pages
-
S-ROM® NOILES™
68 Pages
-
TRI-LOCK® Product Rationale
12 Pages
-
Reclaim Surgical Technique
44 Pages
-
Speed
2 Pages
-
attune
80 Pages
-
HAMMERLOCK® 2
2 Pages
-
DePuy Glenoid Solutions
2 Pages
-
Trauma Solutions. Elbow
4 Pages
-
Polar
4 Pages
-
Alveolar Distractor.
4 Pages
-
Piezoelectric System
4 Pages
-
Air Power Line II
6 Pages
-
LCP Clavicle Hook Plate
4 Pages
-
TruMatch Pin Guides
16 Pages
-
P F N A
8 Pages
-
SKILL, DEDICATION,
16 Pages
-
Orthopaedics. Overview
20 Pages
-
DURALOC
16 Pages
-
Marathon Cemented Cup
20 Pages
-
REEF Surgical Technique
16 Pages
-
MatrixNEURO
8 Pages
-
Anspach XMax
4 Pages
-
Anspach eMax 2 Plus
4 Pages
-
Small Electric Drive
4 Pages
-
Air Pen Drive
4 Pages
-
Colibri II
4 Pages
-
Spine
25 Pages
-
Expert Hindfoot Arthrodesis Nail
48 Pages
-
LCP Distal Fibula Plates
32 Pages
-
TomoFix
60 Pages
-
Expert Tibial Nail PROtect
16 Pages
-
Expert Tibia Nail
84 Pages
-
Sacral Bars
16 Pages
-
Pelvic C-Clamp
20 Pages
-
Low Profile Pelvic System
16 Pages
-
Proximal Femoral (Hook) Plate
24 Pages
-
LCP
24 Pages
-
PFNA
112 Pages
-
HCS 1.5, 2.4, 3.0
36 Pages
-
LCP Wrist Fusion
32 Pages
-
LCP Compact Hand
28 Pages
-
VA-LCP Elbow
48 Pages
-
Distal Radius
44 Pages
-
Olecranon
30 Pages
-
LCP Hook Plate
28 Pages
-
DHP & Olecranon
4 Pages
-
LCP S-A
4 Pages
-
Epoca
4 Pages
-
Philos
32 Pages
-
MultiLoc
68 Pages