- Products
- Catalogs
- News & Trends
- Exhibitions
Catalog excerpts
DRUG-ELUTING PTA BALLOON TECHNOLOGY SPECIFICALLY DESIGNED FOR PERIPHERAL INTERVENTIONS
Open the catalog to page 1FREEWAY ™035 – DRUG-ELUTING PTA BALLOON TECHNOLOGY SPECIFICALLY DESIGNED FOR PERIPHERAL INTERVENTIONS COATING Paclitaxel Paclitaxel is an active ingredient that inhibits the cell replication thus blocking the microtubules decomposition during the metaphase and anaphase stages of mitosis. Shellac is a natural resin composed of shellolic and alleuritic acid. The excellent film forming properties of shellac are used to coat pharmaceutical products and in the food industry. By selectively inhibiting the proliferation of smooth muscle cells, paclitaxel does not influence non-proliferating cells. cell...
Open the catalog to page 2FREEWAY ™035 – DRUG-ELUTING PTA BALLOON TECHNOLOGY SPECIFICALLY DESIGNED FOR PERIPHERAL INTERVENTIONS FREEWAY ™ 035 – COATING CHARACTERISTICS Amorphous Coating The durable non-crystalline bioshell coating homogenously covers the balloon surface and protects the drug from mechanical abrasion and early wash off, resulting in a low paclitaxel blood plasma concentration. level of no toxicity reaction Paclitaxel blood plasma concentrations at 5, 10, 20 and 60 minutes after inflation (120 sec) with FREEWAY DEB.2 Level of toxicity for paclitaxel plasma concentration calculated with a human body surface...
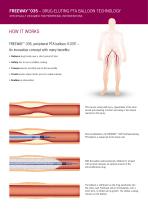
Open the catalog to page 3FREEWAY ™035 – DRUG-ELUTING PTA BALLOON TECHNOLOGY SPECIFICALLY DESIGNED FOR PERIPHERAL INTERVENTIONS HOW IT WORKS FREEWAY™ 035, peripheral PTA balloon 0.035" – An innovative concept with many benefits: • Delivers drug locally over a short period of time • Safety due to non-crystalline coating • Crosses lesions smoothly due to the low profile • Treats lesions where stents are not a viable solution • Enables re-intervention PTA causes vessel wall injury. Hyperplasia of the inner vessel wall resulting in lumen narrowing is the natural reaction to this injury. After predilatation, the FREEWAY ™...
Open the catalog to page 4FREEWAY ™035 – DRUG-ELUTING PTA BALLOON TECHNOLOGY SPECIFICALLY DESIGNED FOR PERIPHERAL INTERVENTIONS FREEWAY ™ 035 PRECLINICAL PROGRAM FREEWAY™ 035 DEB Domestic swine femoral arteries (n = 54) underwent percutaneous overstretch balloon dilation, controlled by optical coherence tomography (OCT). Paclitaxel tissue uptake was measured at 1h, 1 and 3 days. • No delay in endothelialization, no disadvantages in injury and inflammation score compared to standard balloon dilatation (femoral arteries 32 ± 2 days). • FREEWAY ™ 035 DEB demonstrated safety and efficacy in a preclinical model of overstretch...
Open the catalog to page 5FREEWAY ™035 – DRUG-ELUTING PTA BALLOON TECHNOLOGY SPECIFICALLY DESIGNED FOR PERIPHERAL INTERVENTIONS FREEWAY ™ 035 CLINICAL PROGRAM Evidence for the safety and efficacy of the FREEWAY™ 035 DEB better Primary Patency better Primary Patency at 12 months compared to post-stent dilatation with standard balloon at 12 months compared to PTA with standard balloon Study type and focus Study type and focus • randomized multicenter trial in Austria and Germany • randomized study in Austria • 74 patients with in-stent restenosis • 204 patients with de novo or restenotic lesions that needed stent implantation...
Open the catalog to page 6FREEWAY ™035 – DRUG-ELUTING PTA BALLOON TECHNOLOGY SPECIFICALLY DESIGNED FOR PERIPHERAL INTERVENTIONS ADVANCED PRODUCT FEATURES FREEWAY™ 035 DEB – balloons for successful peripheral interventions Dual-lumen catheter shaft Single inflation lumen for fast inflation and deflation times kinking resistant shaft material for crossover procedures • Wide spectrum of balloon catheters for treating long, diffuse lesions – Up to 230 mm balloon length • Elaborated catheter technology with good crossability, trackability and pushability for treatment of diffuse lesions – ual-lumen shaft design with hydrophilic...
Open the catalog to page 7FREEWAY ™035 – DRUG-ELUTING PTA BALLOON TECHNOLOGY SPECIFICALLY DESIGNED FOR PERIPHERAL INTERVENTIONS PRODUCT ORDERING INFORMATION Balloon size diameter × length (mm) Rated burst pressure (atm) Recommended introducer sheath (F) Balloon size diameter × length (mm) Order number Usable catheter length 80 cm Rated burst pressure (atm) Recommended introducer sheath (F) Order number Usable catheter length 135 cm Eurocor Tech GmbH In den Dauen 6a, 53117 Bonn, Germany Phone: +49 (0)228 / 20 15 0-0 Fax: +49 (0)228 / 20 15 0-5 [email protected], eurocor.de Eurocor Tech GmbH is a wholly owned subsidiary of...
Open the catalog to page 8All Eurocor catalogs and technical brochures
- DIOR®
8 Pages