 Website:
Gore
Website:
Gore
Catalog excerpts

Overview GORE® SYNECOR Biomaterial is a unique combination of a macroporous knit of dense, monofilament PTFE fibers and a bioabsorbable copolymer scaffold comprised of polyglycolic acid (PGA) and trimethylene carbonate (TMC) on the parietal surface, and a PGA / TMC nonporous film on the visceral surface. GORE® SYNECOR Biomaterial combines long-term strength with rapid tissue ingrowth and vascularization providing a single-stage durable repair in complex cases. • GORE® BIO-A® Web scaffold facilitates cell infiltration and tissue generation. • A macroporous knit of dense, monofilament PTFE...
Open the catalog to page 2
GORE® SYNECOR Biomaterial is intended for use in the repair of hernias and abdominal wall or thoracic wall soft tissue deficiencies that may require the addition of nonabsorbable reinforcing or bridging material. The material is appropriate for use in open, laparoscopic, or robotic procedures. The combination of the GORE® BIO-A® Web scaffold and the PTFE knit, having a pore size between 1.0 to 3.0 mm, allows for rapid cellular ingrowth and appropriate tissue integration without negatively affecting abdominal wall compliance typically associated with heavyweight polypropylene meshes. Studies...
Open the catalog to page 3
Mean Pore Size and Tissue Response *> '-V -- 4' ■ >- • Nonporous = capsule • 5 and 60 pm pores = vascularized tissue surrounding implant • 700 pm pores = “pseudocapsules” around nodules Typical tissue forming around the polyvinyl alcohol (PVA) implants. The implant is facing up from the bottom and the surrounding tissue is extending down from the top of each micrograph. Small blood vessels near the implant-tissue interface are indicated by small black arrows. Solid nodules of the PVA are indicated by large black arrowheads (original magnification x100). Note the dense tissue structure...
Open the catalog to page 4
Macroporous Knit of Dense, Monofilament Fibers The treatment of ventral hernias with prosthetic devices has reduced recurrence rates but has led to questions concerning infection. Open hernia repair has been associated with infection rates from 3% to 18%.7 Laparoscopic ventral hernia repair has been associated with lower incidence of infection.8 The PTFE knit is designed with a fiber diameter similar to lightweight polypropylene but with the strength of a heavyweight polypropylene. The PTFE knit is designed from a dense, monofilament fiber, similar to polypropylene fibers. The macroporous...
Open the catalog to page 5
Product Use Recommendations GORE® SYNECOR Biomaterial is intended for soft tissue reinforcement. Hospital surgical departments, including general surgery, trauma, and plastics find value in the material and some common applications include: • Laparoscopic ventral hernia repair • Open ventral hernia repair • High risk ventral hernia repair Handling • No pre-soaking of GORE® SYNECOR Biomaterial is needed. • The material is soft and conformable, allowing for easy deployment through the trocar. • Material memory facilitates easy unrolling of the mesh after insertion, for optimal placement. •...
Open the catalog to page 6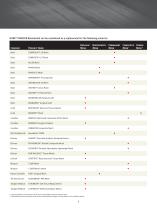
Biologic Biosynthetic Permanent Composite Hybrid Company Product Name Mesh Mesh Mesh Mesh* Mesh** Composite Meshes are permanent mesh with an absorbable visceral protection layer. * Hybrid Meshes have permanent mesh with a bioabsorbable (biologic or biosynthetic) tissue scaffold material.
Open the catalog to page 7
References 1. Klinge U, Klosterhalfen B, Conze J, et al. Modified mesh for hernia repair that is adapted to the physiology of the abdominal wall. European Journal of Surgery 1998;164(12):951-960. 2. Junge K, Klinge U, Prescher A, Giboni P, Niewiera M, Schumpelick V. Elasticity of the anterior abdominal wall and impact for reparation of incisional hernias using mesh implants. Hernia 2001;5(3):113-118. 3. Song C, Alijani A, Frank T, Hanna GB, Cuschieri A. Mechanical properties of the human abdominal wall measured in vivo during insufflation for laparoscopic surgery. Surgical Endoscopy...
Open the catalog to page 8All Gore catalogs and technical brochures
-
Decades of data
2 Pages
-
CONFIGURATIONS
2 Pages
-
Care and Cannulation
9 Pages
-
Introducing
4 Pages
-
ADULT IC CARDIOFORM BROCHURE
8 Pages
-
Brochure Product
4 Pages
-
Pediatric Patient Booklet
36 Pages
-
GORE TIPS Needle
4 Pages
-
GORE® VIABIL®
4 Pages
-
Endovascular
28 Pages
-
GORE-TEX®
4 Pages
-
GORE® DUALMESH® PLUS
6 Pages
-
GORE® SYNECOR Biomaterial
1 Pages
-
GORE-TEX® Suture
6 Pages
-
Cardiothoracic Portfolio
8 Pages
-
Hemodynamic Flow Brochure
4 Pages
-
Product Brochure
4 Pages
-
GORE 3D Imaging System
8 Pages
-
GORE® VIABAHN®
8 Pages
-
GORE® VIABAHN®
24 Pages
-
GORE® SEAMGUARD®
5 Pages
































